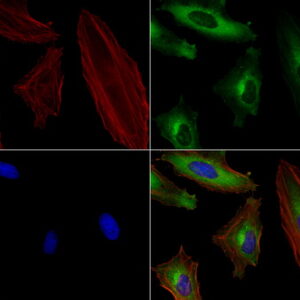
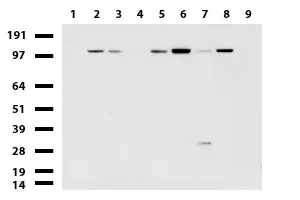
NCAM1 mouse monoclonal antibody, clone UMAB83, 100 uL/ 30 uL
R$12.423,26
| Imunógeno | Fragmento de proteína recombinante humana correspondente aos aminoácidos 20-718 de NCAM1 humano (NP_851996) produzido em E. coli. |
| Aplicações | IHC 1:100, |
| Aplicações2 | IHC |
| Resumo | Este gene codifica uma proteína de adesão celular que é membro da superfamília das imunoglobulinas. A proteína codificada está envolvida em interações célula a célula, bem como interações célula-matriz durante o desenvolvimento e diferenciação. A proteína codificada mostrou estar envolvida no desenvolvimento do sistema nervoso e para células envolvidas na expansão de células T e células dendríticas que desempenham um papel importante na vigilância imunológica. O splicing alternativo resulta em múltiplas variantes de transcrição. |
| Formulação | PBS (pH 7.3) contendo 1% BSA, 50% glicerol e 0.02% sódio azido. |
| Purificação | Purificado a partir de fluidos de ascite de camundongo por cromatografia de afinidade |
| Isotipo | IgG1 |
| Reatividade | Humano |
| Hospedeiro | Camundongo |
| Tamanho | 94.4 kDa |
| Tipo | UltraMAB |
| Concentração | 1.00mg/ml |
” followed by an additional wash with PBS for 5 min. Tissues were subsequently probed with primary antibodies for 1 h at room temperature. The following primary antibodies were used: Anti-CD3 (cat. no. LN10; 1: 50–100 depending on the tissue samples; OriGene Technologies, Inc.), anti-CD56 (cat. no. UMAB83; 1:100; OriGene Technologies, Inc.), anti-CD20 (cat. no. L26; 1:100; Dako; Agilent Technologies, Inc.), anti-TIA-1 (cat. no. 2G9A10F5; 1:100; OriGene Technologies, Inc.), anti-granzyme B (cat. no”
“Histopathology and immunohistochemistrySurgical samples were collected and embedded with paraffin for histological and immunohistochemical analyses. Immunohistochemical analyses for synaptophysin (SYN) (rabbit monoclonal antibody, OriGene, Rockville, MD, USA), chromogranin A (CGA; mouse monoclonal antibody; OriGene), cluster of differentiation (CD56; mouse monoclonal antibody; OriGene), cytokeratin (CK; mouse monoclonal antibody; OriGene), vimentin (VIM; mouse monoclonal antibody; OriGene), S-10”.
” incubation. The primary antibodies used were anti-HA (Roche) as well as, where available, primary antibodies against the native human genes. The primary antibody concentration used was 1 μg/μl for the protein specific antibodies (anti-Ncam1, UMAB83, Origene and anti-Nsf, ab16681, Abcam) and a 1:1000 dilution for the mouse anti-HA. Membranes were incubated with the secondary antibody (GE Healthcare) at a 1:5000 concentration for one hour, followed by signal detection using the Amersham ECL system”.
Produtos relacionados
-
R$12.423,26
Imunógeno Fragmento recombinante expresso em E. coli correspondendo aos aminoácidos 240-390 de CK19 humana Aplicações WB 1:500~1000, IHC 1:50, IF 1:100 Aplicações2 WB, IHC Resumo A queratina 19 é um membro da família da queratina. As queratinas são proteínas de filamentos intermediários responsáveis pela integridade estrutural das células epiteliais e são subdivididas em citoqueratinas e queratinas capilares. As citoqueratinas do tipo I consistem em proteínas ácidas que estão dispostas em pares de cadeias de queratina heterotípicas. Ao contrário de seus parentes relacionados, esta menor citoqueratina ácida conhecida não está emparelhada com uma citoqueratina básica nas células epiteliais. É expresso especificamente na periderme, a camada superficial transitória que envolve a epiderme em desenvolvimento. Formulação PBS (pH 7.3) contendo 1% BSA, 50% glicerol e 0.02% azida sódica. Purificação Purificado a partir de fluidos de ascite de camundongo por cromatografia de afinidade Isotipo IgG1 Reatividade Humano Hospedeiro Camundongo Tamanho 43.9 kDa Tipo UltraMAB Concentração 0.5-1.0 mg/ml -
R$12.423,26
Imunógeno Fragmento de proteína recombinante humana correspondente aos aminoácidos 1544-1830 de DOCK2 humano (NP_004937) produzido em E. coli. Aplicações WB 1:2000, IHC 1:150, Aplicações2 WB,…
-
R$12.423,26
Imunógeno Proteína recombinante humana de comprimento total de CD44 humano (NP_000601) produzida na célula HEK293T. Aplicações IHC 1:500, Aplicações2 IHC Resumo A proteína codificada por…
-
R$12.423,26
Imunógeno Proteína recombinante humana de comprimento total de RAPGEF1 humano (NP_941372) produzida na célula HEK293T. Aplicações IHC 1:100~200, Aplicações2 IHC Resumo Este gene codifica um…